|
Impact of boars’ sperm concentration of ejaculate and rest days between ejaculates on sows’ pregnancy rate
Orlando Díaz 1,2
1 BIOGENESIS Research Group, Universidad de Antioquia Medellín, Colombia 2 MVZ, MsC, Coordinatior of pig farms, Solla S.A. 3 MV. Dr.Sci.Agr Titular Professor, Universidad de Antioquia
This email address is being protected from spambots. You need JavaScript enabled to view it.
Recibido: 26 agosto de 2019, Aprobado: 25 noviembre de 2019, actualizado: 16 diciembre de 2019
DOI: 10.17151/vetzo.2020.14.1.3
ABSTRACT. Introduction: The quality of cooled semen in artificial insemination is important, both for its efficiency in pregnancy rate and for the number of piglets born by litter. Objective: To determine the impact of sperm concentration of ejaculate and rest days between ejaculates on rate of pregnancy and number of piglets born by litter using cooled pig semen. Methods: The study involved the use of 29 ejaculates from 19 boars, which were 229–757 day old, with 5 to 16 rest days between ejaculations, for inseminating 315 sows. The effect of motility, ejaculate volume, ejaculate sperm concentration and diluted sperm concentration, morphology, acrosomal integrity, cell membrane functionality, boar age, rest days between ejaculations, days of semen storage, farrowing, and wean-to-estrus interval were assessed on pregnancy rate and number of piglets born. Linear and logistic regression analyses were performed using SPSS. Results: None of the variables provided a satisfactory explanation for the variation in pregnancy rate and number of piglets born using linear regression. The probability of achieving a pregnancy rate ≥93% was 10.4 times higher when the ejaculates came from boars with ≥12.5 rest days, 9.7 times higher when these ejaculates had a sperm concentration ≥941 million sperm per mL, and 2.7 times higher when the females inseminated were in heat between 4 and 7 days after wean. Conclusion: The ejaculate of boars with more rest days and high sperm concentration had a greater probability of improving the pregnancy rate. The number of piglets remained unaffected.
Key words: pig, fertility, semen
Impacto de la concentración espermática del eyaculado y el tiempo de descanso entre eyaculados de verracos sobre la tasa de preñez en cerdas
RESUMEN. Introducción: La calidad del semen refrigerado en la inseminación artificial es importante, tanto por su eficiencia en la tasa de preñez, como por la cantidad de lechones nacidos por camada. Objetivo: determinar el impacto de la concentración espermática del eyaculado y el descanso entre eyaculaciones sobre la tasa de preñez y la cantidad de lechones nacidos usando semen porcino refrigerado. Métodos: Se usaron 29 eyaculados de 19 verracos entre 229 y 757 días de nacidos, con descansos entre eyaculaciones de cinco a 16 días, para inseminar 315 hembras. Fue evaluado el efecto de la movilidad, el volumen eyaculado, la concentración espermática del eyaculado y del semen diluido, la morfología, la integridad acrosomal, la funcionalidad de la membrana plasmática, la edad del verraco, el tiempo de descanso entre eyaculaciones, los días de almacenamiento del semen, los partos previos y el intervalo entre el destete y el celo sobre la tasa de preñez y sobre la cantidad de lechones nacidos. Se realizaron análisis de regresión lineal y logística con SPSS. Resultados: Ninguna de las variables explicó satisfactoriamente la variación de la tasa de preñez o la cantidad de lechones nacidos por regresión lineal. La probabilidad de obtener una tasa de preñez ≥93% fue 10,4 veces mayor cuando los eyaculados provenían de verracos con 12,5 días o más de descanso, 9,7 veces mayor cuando éstos eyaculados presentaron una concentración espermática ≥941 millones de espermatozoides por ml y 2,7 veces mayor cuando las hembras inseminadas presentaron celo entre cuatro y siete días después de haber sido destetadas. Conclusión: El eyaculado de verracos con mayor descanso y con una concentración espermática alta genera mayor probabilidad de mejorar la tasa de preñez. La cantidad de lechones no es afectada.
Palabras clave: cerdos, fertilidad, semen
Introduction
The number of pregnant females (pregnancy rate) compared to inseminated ones is a better indicator than the number of sows (farrowing rate) for estimating the effect of the semen quality used. This is because of the influence of abortions caused by infectious or environmental factors that are not related to seminal disorders. Although some researchers suggest that embryonic losses can occur due to uncompensable sperm abnormalities (Tardif et al., 1999; Braundmeier & Miller, 2001; Watson & Behan, 2002), it is unclear whether these abnormalities have an effect on pregnancy rate and prolificacy. Prolificacy is the ability to have farrowings with a high number of total piglets born, including living piglets, those that die during early gestation and are mummified, and those that die before or during farrowing. These piglets express the amount of successfully fertilized oocytes that generated viable embryos, with minimal development for achieving a successful implantation in the uterus and forming a fetus (Geisert et al., 2014). Pregnancy rate and prolificacy are concepts directly related to productivity in the swine industry and are dependent on administrative, environmental, and physiological factors, wherein adequate training of the operators, good general condition of the female, optimal time of insemination (Knox & Rodriguez, 2001; Rozeboom et al., 1997; Sandu et al., 2012) and good quality of semen (Pérez-Llano et al., 2001; Dyck et al., 2011; Flowers, 2015) must coincide. Generally, there are in vitro tests for assessing the semen quality, which allow classification of the ejaculates for use in artificial insemination or for disposal (Watson & Behan, 2002; Foxcroft et al., 2008; Saacke, 2008). However, features like sperm concentration of the ejaculate and frequency between ejaculations have not been studied widely during reproductive analyses. Some studies have reported sperm concentration of ejaculates as a general characteristic (Gadea et al., 2004; Smital et al., 2005; Górski et al., 2017; Schulze et al., 2019), and investigations on frequency of ejaculations have reported that the search for maximum intensity of ejaculations had some negative effects on the rate of pregnancy (Frangež et al., 2005; Pruneda et al., 2005; Audet et al., 2009). This study aimed to determine the impact of the sperm concentration of the ejaculate and the frequency between ejaculations on the pregnancy rate of sows using semen refrigerated at 17°C.
Materials and Methods
From November 2017 to May 2018, the rate of pregnancy and number of total piglets born from 29 ejaculates, collected from 19 boars that were 229–757 days old at the time of each ejaculation, were analyzed. The rest time between ejaculates varied between 5 and 16 days. The boars were housed individually in 3 × 2.5 m pens, in a boar semen production center, North of Antioquia, at 2.550 mamsl., with an average temperature of 13.3ºC and annual rainfall of 1.933 mm3. They were fed 3 kg/day of Criacerdos Reproductores® (Solla S.A., Colombia) and water ad libitum. Semen extraction was performed by a qualified operator using the gloved hand technique, where only the sperm-rich fraction (Spz) was collected in a dry glass and with a filter for porcine semen.
A total of 315 females were inseminated, between 0 and 11 farrowings, with semen stored at 17°C from dilution day until use (maximum recorded 9 days). To identify the onset of heat, various heat-check boars (aged >1 year) were exposed in front of the females twice a day, from the second day after weaning until the end of the heat. Nulliparous females were programmed from the third natural cycle of heat in the presence of a heat-check boar. All females were placed in individual gestation cages to be inseminated (cervical insemination) with two doses after the onset of heat: at 0 and 24 h in nulliparous females and at 12 and 24 h in multiparous females. The females that were still in heat 12 h after the second insemination, were inseminated with a third dose of the same ejaculate.
Examination of seminal material: Immediately after obtaining the ejaculate, 10 µl of the semen was extracted to assess sperm motility under a conventional light microscope at 400× magnification, using a slide and a coverslip, both heated to 37°C. The sperm concentration of undiluted semen was estimated in Accuread® photometer (IMV Technologies, France) using a 1:25 dilution of the semen in 2% formol saline. Further, 10 µl of this dilution was used for evaluating sperm morphology on a slide and under a coverslip, using a conventional light microscope at 400× magnification, classifying at least 300 Spz as normal, abnormal (distal cytoplasmic droplet, proximal cytoplasmic droplet, distal midpiece reflex, and head, midpiece, and tail abnormalities), and agglutinates (Díaz et al., 2009). The ejaculate volume was determined in terms of weight using a Scout® Pro electronic scale (OHAUS, Parsippany, New Jersey, United States), assuming that one gm is equivalent to one mL.
Only the ejaculates with ≥80% motility, ≥80% Spz, and normal morphology were diluted to an approximate concentration of 30×l06 Spz per mL in a commercial diluent for porcine semen (Durasperm®, Kruuse, Italy) prepared in distilled water. The diluted ejaculates were packed in bags of 100 mL seminal doses (IMV Technologies, France). One dose of each ejaculate was taken to the Biogenesis laboratory, Faculty of Agricultural Sciences, Universidad de Antioquia, under storage conditions (17°C) similar to the other doses used for inseminations. The sperm motility of the refrigerated semen was verified according to laboratory procedures and the sperm concentration was confirmed with a 1:20 dilution of the refrigerated semen in 2% formol saline solution, using the Bürker-Türk BRAND® counting chamber (Sigma-Aldrich, St. Louis, United States). Plasma membrane functionality was also evaluated for each sample using the sHOST test (short hypoosmotic swelling test, modified from Pérez-Llano et al., 2001) and testing acrosomal membrane integrity by exposure to the fluorochrome FITC-PSA (Lee et al., 2014).
To perform the sHOST assessment, 200 µl of the semen sample was mixed with 600 µl of commercial diluent (Durasperm®) adjusted with distilled water (53 mOsm/kg) (verified in osmometer µOSMETTETM, Model 5004, Precision System INC., United States) for 10 minutes at 37°C in a water bath. Subsequently, a 400 µl aliquot of this mixture was fixed in 400 µl of 2% formol saline solution at room temperature. Then, 10 µl of the fixed mixture was placed on a slide and covered with a coverslip for microscopic examination. At least 100 Spz were visualized and classified using a conventional light microscope (400× magnification). Spz that folded or coiled their flagellum were classified as “viable,” whereas those that remained upright were classified as “non-viable.”
To perform the FITC-PSA assessment, 400 µl of the semen sample mixed with 50 µl of the fluorochrome FITC-PSA (12.5 µg/mL) was incubated at 37°C for 10 minutes in a water bath. Subsequently, 200 µl of this mixture was fixed using 200 µl of 2% formol saline solution at room temperature. From this mixture, 10 µl was deposited on a slide and covered with a coverslip for visualization under a fluorescence microscope at 530 nm wavelength, with a 400× magnification in dark conditions. At least 100 Spz for each sample were classified as “non-viable” (with acrosome reaction) for those with some degree of fluorescence (green) in the acrosomal region and as “viable” (complete acrosome) for those that did not fluoresce.
Obtaining field information and statistical analyses: The insemination results were extracted into a Microsoft® Excel® 2016 spreadsheet using PigKnows LLC Version 2.11.0 (Pig Farming Data Management System, United States). The analyses were performed using SPSS Version 25.0 (IBM Corp, 2017, IBM SPSS Statistic for Windows, Armonk, NY). The dependent variables were pregnancy rate (proportion of inseminated females that did not repeat heat until 25 days after insemination) and total piglets born. The independent variables were motility, morphology, ejaculate volume, sperm concentration, cell membrane functionality, acrosomal integrity, boar’s age at the time of collection, rest time between collections, days of semen storage, previous farrowings, and interval of time between weaning and the heat onset. The effect of the independent variables on the dependent variables was evaluated using multiple linear regression. Through descriptive analysis, the average values were considered to categorize the following variables into 0 and l: pregnancy rate and total number of piglets born; and to perform a multiple binary logistic regression analysis, the following independent variables were categorized into 0 and l according to their averages: previous farrowings, ejaculate volume, sperm concentration of the ejaculate, sperm concentration of diluted semen, boar’s age at the time of collection, rest time between collections, and weaning-to-heat time interval. P < 0.05 was considered statistically significant.
Results and Discussion
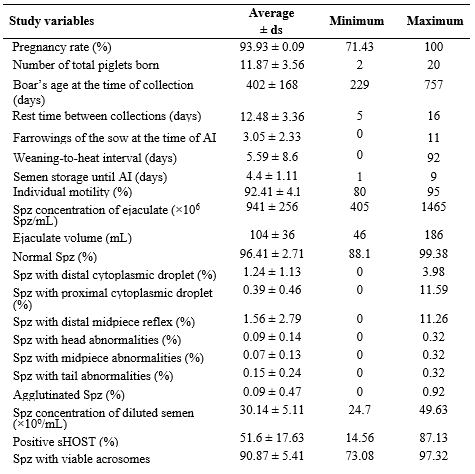
The variation in pregnancy rate or in the number of total piglets born could not be explained satisfactorily using any linear regression model that included the studied variables. The coefficient of determination of the best linear regression model was R2 = 0.316, including different confounding variables. Additionally, the assumptions of normality, homoscedasticity, and non-collinearity of the residuals were not fulfilled (data not shown). The descriptive analysis of the pregnancy rate and the number of total piglets born has been depicted in table 1, along with other reproductive parameters and semen quality.
Table 1. Statistical description of the analyzed reproductive parameters
Sd: Standard deviation. Spz: Sperm. AI: Artificial insemination. sHOST: Cell membrane functionality test.
In a Benchmark (ACP, 2016), the Asociación Colombiana de Porcicultores reported in 2016 an average of 12.81 total piglets born and an 88.63% farrowing rate, which was reported for 14,838 sows from 54 Colombian farms that voluntarily shared their data. In this study, fewer total piglets born were reported than those in the Benchmark (ACP, 2016); additionally, the pregnancy rate between the two could not be compared. The productive information of all Colombian pig farms has not been consolidated and much of the information remains unknown.
The pregnancy rate was categorized, considering the average value, into 0 for pregnancy percentage <93% (n = 96) and into l for pregnancy percentage ≥93% (n = 202). The number of total piglets born was categorized as 0 when the litter size at each parturition was <11.8 piglets (n = 79) and as 1 when it was ≥11.8 (n = 139). The number of previous parturitions of the sows, sperm concentration of the ejaculate and diluted semen, age at the time of collection, rest time between ejaculations, weaning-to-estrus interval, and volume of the ejaculate were also categorized according to their average values.
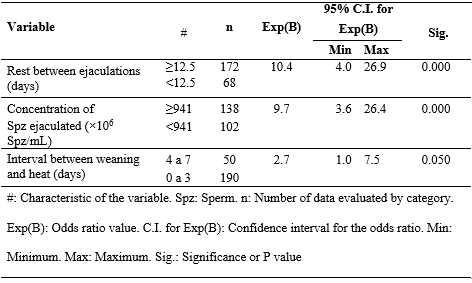
The probability of obtaining a pregnancy rate ≥93% was 10.4 times higher when the ejaculates came from boars with a rest period of ≥12.5 days between ejaculations, 9.7 times higher when these ejaculates had a sperm concentration ≥94l × l06 Spz/mL, and 2.7 times higher when the inseminated females came into heat between 4 and 7 days after weaning (P < 0.01). Table 2 shows the odds ratio of the multiple binary logistic regression model on the pregnancy rate. The categories of previous farrowings, ejaculate volume, sperm concentration of diluted semen, and age at the time of collection did not have a statistically significant impact on the pregnancy rate categories. The probability of having litters with ≥11.8 total piglets born was not affected by any of the analyzed categories.
Table 2. Multiple binary logistic regression statistics of the categories associated with a ≥93% pregnancy rate.
#: Characteristic of the variable. Spz: Sperm. n: Number of data evaluated by category. Exp(B): Odds ratio value. C.I. for Exp(B): Confidence interval for the odds ratio. Min: Minimum. Max: Maximum. Sig.: Significance or P value
Regarding the rest time between ejaculations, different investigations have approached the subject focusing on the maximum possible frequency, perhaps seeking to determine the maximum use of boars; as a consequence, there was a decrease in the volume and sperm concentration of the ejaculate, as well as in the pregnancy rate (Frangež et al., 2005; Pruneda et al., 2005; Audet et al., 2009). Ejaculation interval >10 days increases sperm concentration and ejaculate volume, as well as the number of functional sperm, compared to intervals <10 days (Wolf & Smital, 2009). The time required for sperm to pass through the porcine epididymis (50–100 m in length) is 9–11.8 days (França & Cardoso, 1998; França et al., 2005) and the fact that the tail of the epididymis serves as a sperm reservoir (Dacheux et al., 2005; 2016) could be a reason for the above. If the resting period is similar to the storage capacity of the epididymis tail, the Spz would have a longer exposure time to the epididymis environment between each ejaculation of the boar.
Ruiz-Sánchez et al. (2006) reported that boar ejaculate is released in fractions and that it is possible to differentiate the sperm-rich fraction from the sperm-deprived fraction. Different averages of sperm concentration (millions of Spz/mL) have been reported, such as 316 ± 147 (Schulze et al., 2019), 289 ± 54 (Frangež et al., 2005), 413 ± 105 (Smital et al., 2005), and 497 ± 85 (Górski et al., 2017), but without any relationship with fertility. However, Gadea et al. (2004) reported an average sperm concentration of 882 ± 26 × 106 Spz/mL in ejaculates that generated a farrowing rate >85%, which was higher than the ejaculates with 779 ± 25 × 106 Spz/mL sperm concentration and a farrowing rate ≤85%. This finding was similar to our results, where higher sperm concentration resulted in higher pregnancy rate.
The porcine ejaculate comprises Spz and fluids from the testicle, epididymis, and accessory sex glands (Rodríguez-Martínez et al., 2011; López Rodríguez et al., 2013). The reported sperm concentration of the ejaculates in studies by Schulze et al. (2019), Frangež et al. (2005), Smital et al. (2005), and Górski et al. (2017) can be caused by the collection of a greater amount of secretions from the accessory sex glands during ejaculation, compared to that reported by Gadea et al. (2004), where the collection could be of a lower quantity of these secretions. However, it is not sufficiently clear which fraction of the ejaculate was collected and used.
Barranco et al. (2015) found a positive correlation of fertility and litter size with the total antioxidant capacity of ejaculates, which is associated with the antioxidant capacity of the respective ejaculate fractions. Vilagran et al. (2015) demonstrated that quantitative differences in the composition of seminal plasma greatly affect the variation in resistance to sperm freezing, similar to that reported by Peña et al. (2006), based on the differences of ejaculate fractions. The antioxidant properties of porcine ejaculate could be influenced by the fluid secreted from the seminal vesicles (Caballero et al., 2008) or from the epididymal fluid (Dacheux et al., 2005; Souza et al., 2017). Proteomic analyses of the ejaculate fractions could help explain the variation in pregnancy rate and litter size, generating a range of possibilities for future investigations.
In this study, the higher probability of generating a pregnancy rate ≥93% for sows with a weaning-to-estrus interval of 4–7 days could be related to the findings of Knox & Rodriguez (2001), where sows that came into heat 4 days after weaning had a higher number of complete ovulations than those that came into heat before 4 days.
According to the findings of this study, it is possible to make the ejaculation frequency of boars more flexible, up to 16 days, aiding the maximal use of animals with greater genetic merit. It is necessary to consider that the experimentation conditions were subject to a productive system without total control of the environment, which could influence the reproductive results. Hence, it is possible to conduct investigations with controlled environments. Additionally, it is feasible to establish a research field investigating proteomics of the ejaculate fractions on pregnancy rate and number of total piglets born using artificial insemination of sows with refrigerated semen.
Conclusions
The probability of improving reproductive results in pig farming can be calculated through the analysis of information and, in particular, can depend on variables that are not routinely analyzed, such as interval between ejaculations or sperm concentration of the ejaculate. When the routine variables intended to assess seminal quality (sperm motility and morphology) meet the acceptance criteria, it is customary to use the ejaculate for artificial insemination. However, this causes variability in the results, which may be associated with factors of the epididymis, fractions of the ejaculate, pending analysis, and field results owing to the lack of efficient articulation between the University, the private companies, and the Colombian state.
References
ACP, Asociación Colombiana de porcicultores. Análisis de la industria porcícola en Colombia. Porcicultura Colombiana, 208° ed. v.4, V. 12, p. 36, 2016.
Audet, I.; Bérubé, N.; Bailey, J.L. et al. Effects of dietary vitamin supplementation and semen collection frequency on hormonal profile during ejaculation in the boar. Theriogenology, V. 71, 334-341, 2009.
Barranco, I.; Tvarijonaviciute, A.; Perez-Patiño, C. et al. High total antioxidant capacity of the porcine seminal plasma (SP-TAC) relates to sperm survival and fertility. Scientific Reports, V. 5, 18538, p. 9, 2015. Available at: https://www.nature.com/articles/srep18538. Accession date: 01/19/2017.
Braundmeier, A.G.; Miller, D.J. The search is on: finding accurate molecular markers of male fertility. Journal of Dairy Science, V. 84, p. 1915-1925, 2001.
Caballero, I.; Vazquez, J.M.; García, E.M. et al. Major proteins of boar seminal plasma as a tool for biotechnological preservation of spermatozoa. Theriogenology, V. 70, p. 1352-1355, 2008.
Dacheux, J.L.; Castella, S.; Gatti, J.L.; Dacheux, F. Epididymal cell secretory activities and the role of proteins in boar sperm maturation. Theriogenology, V. 63, p. 319-341, 2005.
Dacheux, J.L.; Dacheux, F.; Druart, X. Epididymal protein markers and fertility. Animal Reproduction Science, V. 169, p. 76-87, 2016.
Díaz, O.; Valencia-Mejía, J.G.; Gómez-Londoño, G. et al. Evaluación de la integridad acrosomal y la funcionalidad bioquímica de la membrana plasmática en cerdos reproductores con gotas citoplásmicas persistentes. Revista Científica, FCV-LUZ, V. 19, p. 500-505, 2009.
Dyck, M.K.; Foxcroft, G.R.; Novak, S. et al. Biological markers of boar fertility. Reproduction in Domestics Animals, V. 46 Supplement 2, p. 55-58, 2011.
Foxcroft, G.R.; Dyck, M.K.; Ruiz-Sanchez, A. et al. Identifying useable semen. Theriogenology, V. 70, p. 1324-1336, 2008.
França, L.R.; Avelar, G.F.; Almeida, F.F.L. Spermatogenesis and sperm transit through the epididymis in mammals with emphasis on pigs. Theriogenology, V. 63, p. 300-318, 2005.
França, L.R.; Cardoso, F.M. Duration of spermatogenesis and sperm transit time through the epididymis in the Piau boar. Tissue and Cell, V. 30, p. 573-582, 1998.
Frangež, R.; Gider, T.; Kosec, M. Frequency of boar ejaculate collection and its influence on semen quality, pregnancy rate and litter size. Veterinaria Brno, V. 74, p. 265-273, 2005.
Gadea, J.; Sellés, E.; Marco, M.A. The predictive value of porcine seminal parameters on fertility outcome under commercial conditions. Reproduction in Domestic Animals = Zuchthygiene, V. 39, p. 303-308, 2004.
Geisert, R.D.; Lucy, M.C.; Whyte, J.J. et al. Cytokines from the pig conceptus: roles in conceptus development in pigs. Journal of Animal Science and Biotechnology, V. 5, p. 51, p.l-12, 2014.
Górski, K.; Kondracki, S.; Wysokínska, A. Ejaculate traits and sperm morphology depending on ejaculate volume in Duroc boars. Journal of Veterinary Research, V. 61, p. 121-125, 2017.
Knox, R.V.; Rodriguez Zas, S.L. Factors influencing estrus and ovulation in weaned sows as determined by transrectal ultrasound. Journal of Animal Science, V. 79, p. 2957-2963, 2001.
Lee, W.Y.; Lee, R.; Kim, H.C. et al. Pig Spermatozoa Defect in Acrosome Formation Caused Poor Motion Parameters and Fertilization Failure through Artificial Insemination and In vitro Fertilization. Asian-Australasian Journal of Animal Sciences, V. 27, p. 1417-1425, 2014.
López Rodríguez, A.; Rijsselaere, T.; Beek, J. et al. Boar seminal plasma components and their relation with semen quality. Systems Biology in Reproductive Medicine, V. 59, p. 5-12, 2013.
Peña, F.J.; Saravia, F.; Núñez-Martínez, I. et al. Do different portions of the boar ejaculate vary in their ability to sustain cryopreservation? Animal Reproduction Science, V. 93, p. 101-113, 2006.
Pérez-Llano, B.; Lorenzo, J.L.; Yenes, P. et al. A short hypoosmotic swelling test for the prediction of boar sperm fertility. Theriogenology, V. 56, p. 387-398, 2001.
Pruneda, A.; Pinart, E.; Dolors Briz, M.D. et al. Effects of a high semen-collection frequency on the quality of sperm from ejaculates and from six epididymal regions in boars. Theriogenology, V. 63, p. 2219-2232, 2005.
Rodríguez-Martínez, H.; Kvist, U.; Ernerudh, J. et al. Seminal plasma proteins: what role do they play? American Journal of Reproductive Immunology, V. 66, No. l Supplement 1, p. 11-22, 2011.
Rozeboom, K.J.; Troedsson, M.H.T.; Shurson, G.C. et al. Late estrus or metestrus insemination after estrual inseminations decreases farrowing rate and litter size in swine. Journal of Animal Science, V. 75, p. 2323-2327, 1997.
Ruiz-Sánchez, A.L.; O’Donoghue, R.; Novak, S. et al. The predictive value of routine semen evaluation and IVF technology for determining relative boar fertility. Theriogenology, V. 66, p. 736-748, 2006.
Saacke, R.G. Sperm morphology: its relevance to compensable and uncompensable traits in semen. Theriogenology, V. 70, p. 473-478, 2008.
Sandu, M.; Mantea, S.; Ipate, I. et al. Study upon the moment of ovulation in sows to establish the optimum moment for semen inoculation. Animal Science and Biotechnologies, V. 45, p. 346-348, 2012.
Schulze, M.; Jakop, U.; Jung, M.; Cabezón, F. Influences on thermo-resistance of boar spermatozoa. Theriogenology, V. 127, p. 15-20, 2019.
Smital, J.; Wolf, J.; De Sousa, L.L. Estimation of genetic parameters of semen characteristics and reproductive traits in AI boars. Animal Reproduction Science, V. 86, p. 119-130, 2005.
Souza, A.P.Bd; Schorr-Lenz, Â.M.; Lucca, F.; Bustamante, I.C. The epididymis and its role on sperm quality and male fertility. Animal Reproduction, V. 14, 1234-1244, 2017.
Tardif, S.; Laforest, J.P.; Cormier, N.; Bailey, J.L. The importance of porcine sperm parameters on fertility in vivo. Theriogenology, V. 52, p. 447-459, 1999.
Vilagran, I.; Yeste, M.; Sancho, S. et al. Comparative analysis of boar seminal plasma proteome from different freezability ejaculates and identification of Fibronectin 1 as sperm freezability marker. Andrology, V. 3, p. 345-356, 2015.
Watson, P.F.; Behan, J.R. Intrauterine insemination of sows with reduced sperm numbers: results of a commercially based field trial. Theriogenology, V. 57, p. 1683-1693, 2002.
Wolf, J.; Smital, J. Quantification of factors affecting semen traits in artificial insemination boars from animal model analyses. Journal of Animal Science, V. 87, p. 1620-1627, 2009.
Como citar: Díaz O., Olivera-Ange M. Impact of boars’ sperm concentration of ejaculate and rest days between ejaculates on sows’ pregnancy rate. Revista Veterinaria y Zootecnia. n, v. 14, n. 1, p. 00-00, 2020. http://vetzootec.ucaldas.edu.co/index.php/component/content/article?id=284. DOI: 10.17151/vetzo.2020.14.1.3
Esta obra está bajo una Licencia de Creative Commons Reconocimiento CC BY
|